 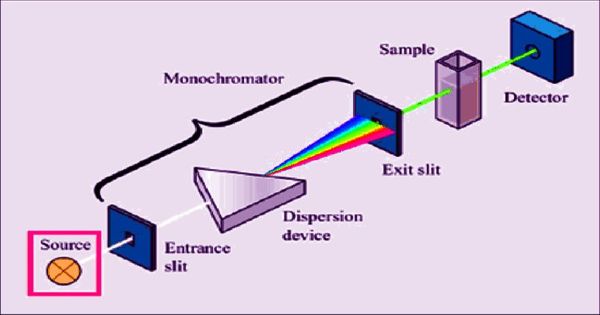
In gas phase spectroscopy, the fine structure afforded by these factors can be discerned, but in solution-state spectroscopy, the differences in molecular microenvironments further broaden the structure to give smooth bands. The main factors that cause broadening of the spectral line into an absorption band are the distributions of vibrational and rotational energies of the molecules in the sample (and also those of their excited states). Types of absorption band Electronic transitionsĮlectronic transitions mainly take place at energies corresponding to the UV and visible part of the spectrum. There are many mechanisms by which line broadening into bands can occur, including: All the same, in many cases it is convenient to assume that a spectral band is an easily modeled shape such as a Gaussian or Lorentzian. Thus, rather than an infinitely narrow absorbance line in the spectrum, a band is observed - a range within which a particular transition may take place under the right conditions.Ībsorption band characteristics Band shapeĪ wide variety of band shapes exist, and the analysis of the band shape can be used to determine information about the transition that casues it. For this reason each transiton happens at a slightly different wavelength in different molecules. They will be moving differently, vibrating and rotating differently, and have different neighbors. However, not all molecules are exactly the same, even those of the same substance. This is the origin of the absorption spectrum. Thus, when light of one of these specific wavelengths interacts with a molecule, it can absorb it while other wavelengths pass. Each of these steps corresponds to a particular energy, which may be represented as a wavelength of light (or a spectral line). Therefore, in order for a substance to change its energy it must do so in a series of "steps" conforming to the allowed states that it may exist in. 4 Absorption bands of interest to the atmospheric physicistĪccording to quantum theory, atoms and molecules can only hold certain defined quantities of energy, or exist is specific states. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
